
A HIGHER STANDARD
From inception, Red Mesa remains dedicated to producing consistently safe, high-quality, hemp-derived CBD and CBG raw materials, while delivering exceptional customer service and support.
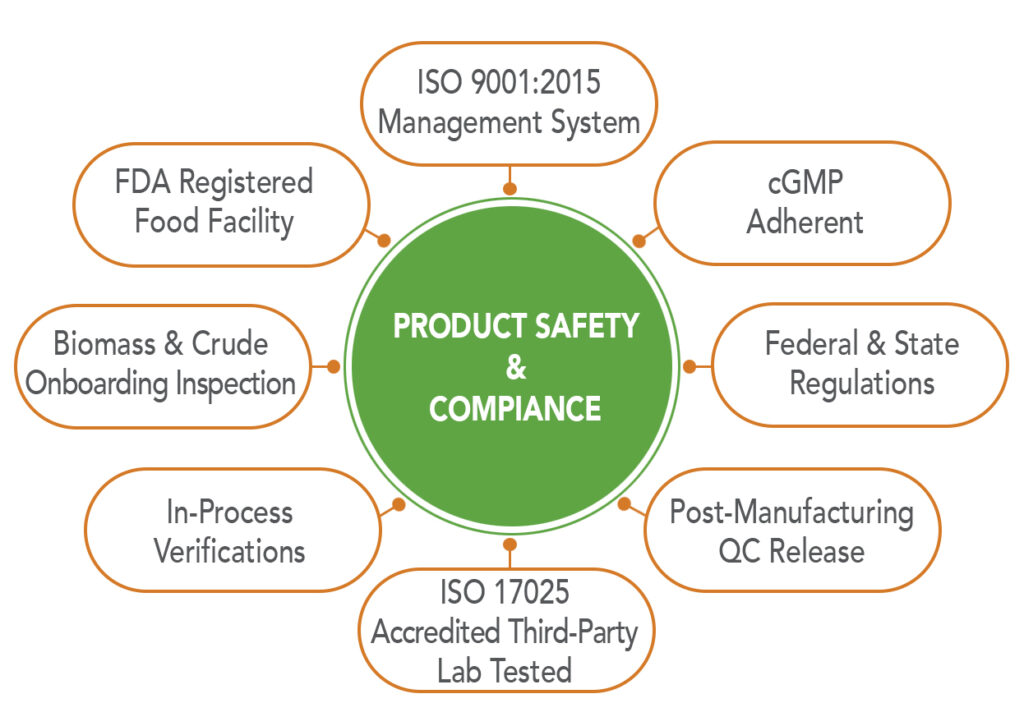
Red Mesa set high standards and self-imposed stringent operational guidelines greater than required by the industry but central to being an ethical partner in your supply chain. From day one, the company’s foundation was built upon the quality management principles of ISO 9001:2015 and adherence to Current Good Manufacturing Practices (cGMP) to ensure integrity in operations and manufacturing processes, cleanliness throughout the facility and compliance with safety regulations.
Throughout the production process, our team members are committed to adhering to both federal and Utah state regulations, in addition to internal data-driven requirements, to ensure professionalism, reliability and trustworthiness.
ISO 9001:2015 
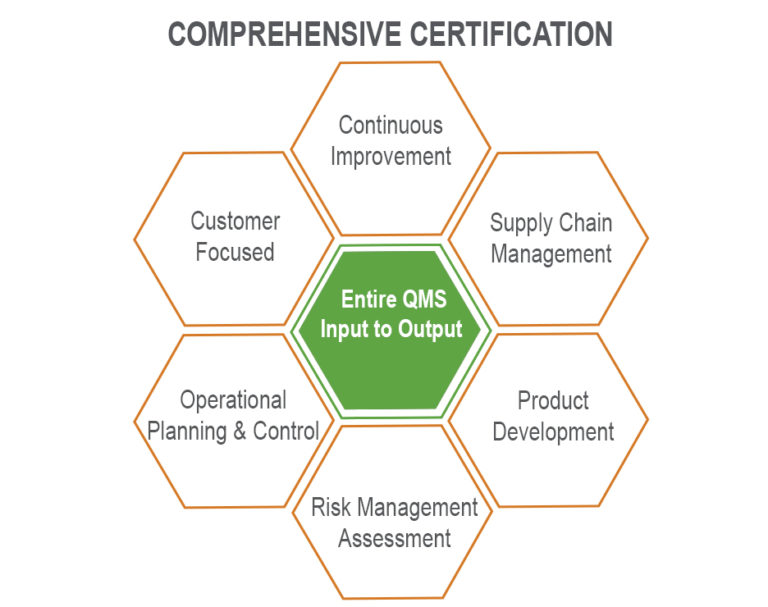
Red Mesa’s Quality Management System (QMS) is ISO 9001:2015 certified. The scope of our QMS supports the end-to-end processing of industrial hemp under Utah’s Industrial Hemp Program for Processors. Our ISO 9001:2015 certification serves as our daily guide and reinforces our focus and commitment to continuously improve to achieve optimal quality and safety.
To maintain certification, we abide by our Quality System Risk Management Protocol, a key component of ISO 9001:2015, to manage the business and ensure customer satisfaction. Additionally, we prioritize ongoing product development and supply chain management to support timely deliveries of consistently high-quality products. (read more)
ADHERENCE TO CGMP REGULATIONS
The company-wide integration of our Quality Management System ensures our Current Good Manufacturing Practices (cGMP) adherence and prioritization of the reliability and consistency of product, facility cleanliness and customer safety. Every step of our manufacturing process is documented, with all materials used in production being traceable throughout. (read more)
cGMP requirements include:
- An established Quality Management System.
- Documented procedures that seek to ensure consistency of operations.
- Detailed record-keeping, ensuring end-to-end traceability.
- Supply chain program to approve and monitor vendors.
- Controls for production, sanitization, hygiene, equipment, packaging & labeling, warehousing and distribution.

STATE OF UTAH REGULATORY REQUIREMENTS
As a licensed processor of industrial hemp and operating in the state of Utah, we are required to adhere to both Dietary Supplements (21 CFR Part 111) and Food Regulations (21 CFR Part 117). Consequently, we process and refine hemp-derived CBD and CBG raw materials under broader regulatory authority and scrutiny.
To ensure no traces of harmful chemicals or organisms are present in our cannabinoid raw ingredients, Red Mesa is subjected to some of the strictest testing parameters, requiring the lowest detection among Utah’s comprehensive list. While conducting business as a CBD manufacturer in Utah adds another layer of stringent regulations, we embrace our location as one of the few states currently implementing FDA requirements for cGMP in preparation for finalized regulations. While it remains uncertain how the FDA will ultimately classify CBD, we are prepared to move forward with active pharmaceutical ingredients if the FDA decides to categorize it as such. (read more).
IN-HOUSE LABORATORY FACILITY
Our in-house lab utilizes HPLC (high-performance liquid chromatography), among other equipment, to perform small-scale R&D to relay accurate data for daily operations. The lab’s proximity to the manufacturing floor allows for routine analytical testing throughout the manufacturing process to support decision-making regarding consistency, production controls, and process optimization.
ISO 17025 ACCREDITED THIRD-PARTY LAB CoAs
The state of Utah requires tests to all of Utah’s requirements including cannabinoid potency, heavy metals, residual solvents, pesticide residue, foreign material and other microbial contaminants. (read more)
Each batch-specific CoA (Certificate of Analysis) is accessed via product label QR code and will report the following:
- Cannabinoid Profile
- Pesticides
- Residual Solvents
- Microbial Analysis
- Mycotoxins
- Heavy Metals
- Foreign Material
PRODUCT SPECIFICATIONS SHEETS – YOUR GUARANTEE
Product Specification Sheets, available for download, detail guaranteed minimum cannabinoid concentration levels and product characteristics in support of the Red Mesa Standard and our strongly held belief – the promises we speak of are the results we deliver. Each Product Specification Sheet provides general information regarding material usage, restrictions, handling, storage and shelf life, as well as clearly defined characteristics you can consistently expect upon each order.

